 Rates of zinc and trace metal release from dissolving sphalerite at pH 2.0-4. The Mn distribution in high Mn samples is nearly independent of pH and sphalerite dissolution at pH 2.0 but shows a dependence on these two parameters at higher pH (3.0-4.0). The Mn distributions suggest an increase in the rate of Mn release relative to sphalerite dissolution occurs in low Mn samples as pH increases. The distribution of Cu is similar to that of Fe but is the most-dependent of all metals on its mole fraction ratio (Zn:Cu) in sphalerite. The Pb distribution shows no strong relation to sphalerite dissolution and appears to be controlled by pH-dependent solubility, most likely related to trace amounts of galena. These two metals are likely to be more amenable to usage in predictive acid dissolution models because of this behavior. Results indicate that the distributions of Fe and Cd are directly related to the rate of sphalerite dissolution throughout the reaction at pH 3.0 and 4.0 because these two elements substitute readily into sphalerite. Trace metal release rates are generally 1-5 orders of magnitude slower than the Zn or Fe rates. At pH 3.0 and 4.0, release rates show less variation and appear less dependent on the metal abundance in the solid. Explanation: Zinc (Zn), chemical element, a low-melting metal of Group 12 (IIb, or zinc group) of the periodic table, that is essential to life and is one. The largest variations in metal release rates occur at pH 2.0. Zinc and Fe exhibit the fastest rates of release, Mn and Pb have intermediate rates of release, and Cd and Cu show the slowest rates of release. Over the pH range 2.0-4.0, and for similar extent of reaction (reaction time), sphalerite composition, and surface area, the rates of release of Zn, Fe, Cd, Cu, Mn and Pb from sphalerite generally increase with lower pH. Nonoxidative sphalerite dissolution (mediated by H+) is characterized by a "fast" stage in the first 24-30 h, followed by a "slow" stage for the remainder of the reaction. Departures from the ideal Dtr of 1.0 suggest that some trace metals may be released via different pathways or that other processes (e.g., adsorption, solubility of trace minerals such as galena) affect the observed concentration of metals. The distribution was used to determine some controls on metal release during the dissolution of sphalerite. This coefficient describes the relationship of the sphalerite dissolution rate to the trace metal mole fraction in the solid and its aqueous concentration. The release (solubilization) of trace metals from dissolving sphalerite to the aqueous phase can be characterized by a kinetic distribution coefficient, (Dtr), which is defined as, where R is the trace metal or Zn release rate, and X is the mole fraction of the trace metal or Zn in sphalerite. Suggest that the formation timescale of the thick disk is ~ 1-3 Gyr.High-Fe and low-Fe sphalerite samples were reacted under controlled pH conditions to determine nonoxidative rates of release of Zn and trace metals from the solid-phase. We also show the abundance ratios and the metallicityĭistribution functions of the Galactic bulge, halo, and thick disk. < -1$, and the observed trend at the lower metallicity can be explainedīy the energy effect. (Cr, Mn, Co, and Ni) are consistent with the observed mean values at -2.5 < Reproduced by the metallicity effect on nucleosynthesis. Observed decrease in the odd-Z elements (Na, Al, and Cu) toward low is The observedĪbundance of Zn ( ~ 0) can be explained only by the high energyĮxplosion models, which requires a large contribution of hypernovae. 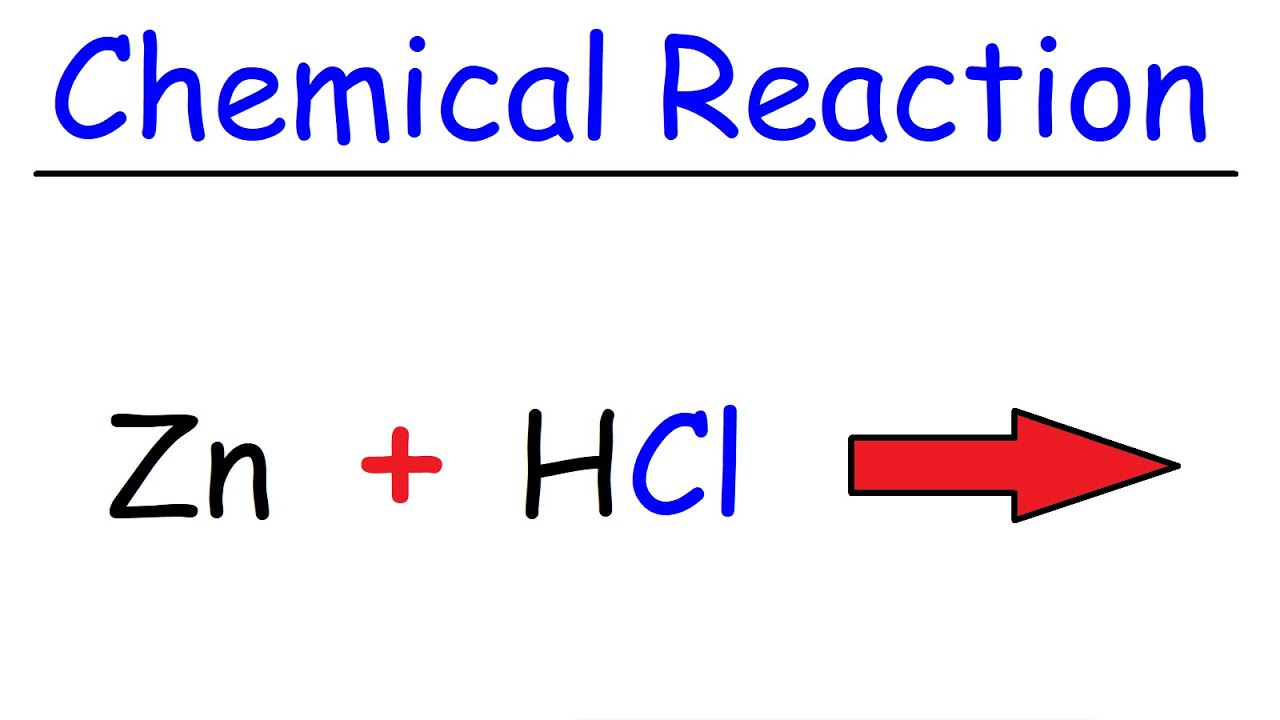
Show a plateau at < -1, while Ti is underabundant overall. Among the alpha-elements, O, Mg, Si, S, and Ca The elemental abundance ratios are in goodĪgreement with observations. Our yields areĬalculated for wide ranges of metallicity (Z=0-Z_\odot) and the explosionĮnergy (normal supernovae and hypernovae), based on the light curve and spectraįitting of individual supernovae. Solar neighborhood adopting our new nucleosynthesis yields. #ZN ELEMENT PH PDF#Kobayashi and 4 other authors Download PDF Abstract: We calculate the evolution of heavy element abundances from C to Zn in the Download a PDF of the paper titled Galactic chemical evolution: Carbon through Zinc, by C. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
